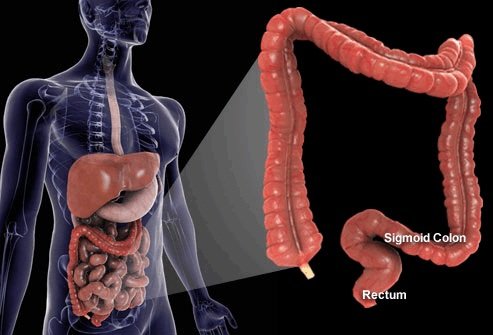
Training
Course Overview
This comprehensive training program covers the installation, operation, maintenance, and regulatory compliance of Clearwater Colon Hydrotherapy equipment. It also includes practical procedures, FDA guidelines, and business setup essentials for your colon prep department.
Equipment Training
Learn step-by-step procedures for: Installation of colon hydrotherapy systems Filter system maintenance and filter replacement Disinfection protocols for equipment hygiene Routine equipment maintenance for optimal performance Proper use of disposable speculums (Bulb and Straight styles)
Regulatory Compliance: FDA Guidelines
| Regulation | Description |
|---|---|
| 21 CFR 801.109 | Adequate Directions for Use & Prescription Statement |
| 21 CFR 876.5220 | Indication for Use Statement |
Call about courses to become a certified colon hydrotherapist
Business & Clinical Knowledge
Contraindications for colon hydrotherapy
Business guidelines for setting up a compliant colon prep department
How to complete FDA 3500A reports for serious adverse events
Colonic Procedure Training
Hands-on instruction in:
Proper technique and application of colon hydrotherapy
Best practices for client comfort and safety
Troubleshooting common procedural issues
Manufacturer Equipment Department Training
Gain deep insight into the technology and engineering behind the systems:
History of Clearwater equipment and manufacturing standards
FDA manufacturing requirements and traceability protocols
Component breakdown and system functionality
Understanding colon pressure vs. water pressure and flow
Why FDA mandates colon pressure monitoring (not water pressure)
How flow controls impact pressure gauges and why this is a critical design consideration